|
Other transport pathways (eg, CO 2, amino acids, glucose, urea, etc) are not shown. NMDAR, the multicomponent N-methyl d-aspartate receptor participating in calcium regulation in erythroid cells, is also shown. Active transporters are the calcium ATPase, encoded by ATP2B4, and sodium-potassium ATPase, the multicomponent sodium pump composed of α1 and α3 isoforms, β1, β2, and β3 isoforms, and the γ modulator. Other erythrocyte membrane channels are the aquaporins, water channels encoded by AQP1 and AQP3, and KCNN4 encoding the Gardos channel, a calcium-activated potassium channel. PIEZO1 is a mechano-activated channel of the erythrocyte membrane. Gradient-driven leak pathways, not otherwise identified and indicated by the arrows, also exist in mature erythrocytes. Gradient-driven transport pathways include AE1: the anion exchange protein 1, band 3, encoded by SLC4A1 NKCC1 encoding Na-K-Cl cotransporter 1 KCC: K-Cl cotransporters of the family of chloride-cation cotransporters VDAC: voltage-dependent anion channels NHE1 encoding the sodium/hydrogen exchanger 1 and VRAC: voltage-regulated anion channels, encoded in part by SWELL1 ( LRRC8A). Ion transport pathways of the human erythrocyte are shown. Model of ion transport pathways of the human erythrocyte. 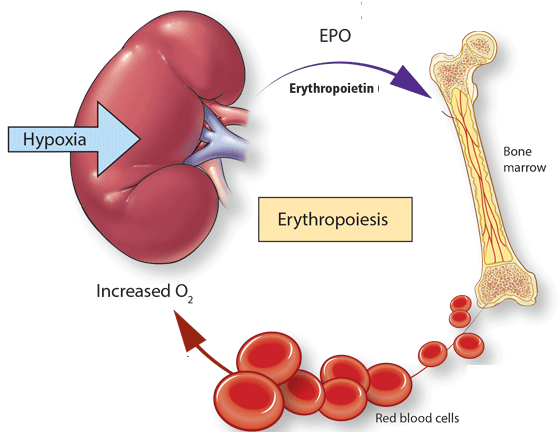
These mechanisms will serve as a paradigm for other cells and may reveal new therapeutic targets for disease prevention and treatment beyond the erythrocyte. Understanding the pathways regulating erythrocyte water and solute content may reveal innovative strategies to maintain normal volume in disorders associated with primary or secondary cellular dehydration. Secondary disorders of erythrocyte hydration include sickle cell disease, thalassemia, hemoglobin CC, and hereditary spherocytosis, where cellular dehydration may be a significant contributor to disease pathology and clinical complications. They have implicated roles for several proteins, PIEZO1, a mammalian mechanosensory protein GLUT1, the glucose transporter SLC4A1, the anion transporter RhAG, the Rh-associated glycoprotein KCNN4, the Gardos channel and ABCB6, an adenosine triphosphate–binding cassette family member, in the maintenance of erythrocyte volume homeostasis. Recent studies have revealed that these disorders are also marked by genetic heterogeneity. 
These disorders are marked by clinical, laboratory, and physiologic heterogeneity. Primary disorders that perturb volume homeostasis jeopardize the erythrocyte and may lead to its premature destruction. This allows the erythrocyte to maintain a narrow range of cell hemoglobin concentration, a process critical for normal red blood cell function and survival. The erythrocyte contains a network of pathways that regulate salt and water content in the face of extracellular and intracellular osmotic perturbations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
